Health Proof Helsinki® ecosystem offers preclinical and clinical testing and research services with expertise
Health Proof Helsinki® (HPH) is an ecosystem, in which Metropolia Proof Health®, together with HUS and the City of Helsinki, are in the forefront of European research, innovation and development. Health Proof Helsinki has enabled numerous joint research projects, generated advanced analytics based on existing datasets, and served as a platform for launching and participating in international research collaborations.
Metropolia Proof Health®
Metropolia Proof Health® (MPH) is a coordinator of the ecosystem and one of the three actors in the ecosystem. Metropolia Proof Health provides advanced preclinical and clinical testing and verification environments for various users across all the stages required to develop health innovations.
At Myllypuro campus in Helsinki Finland we offer modern facilities for companies, research institutes, and public sector organizations.
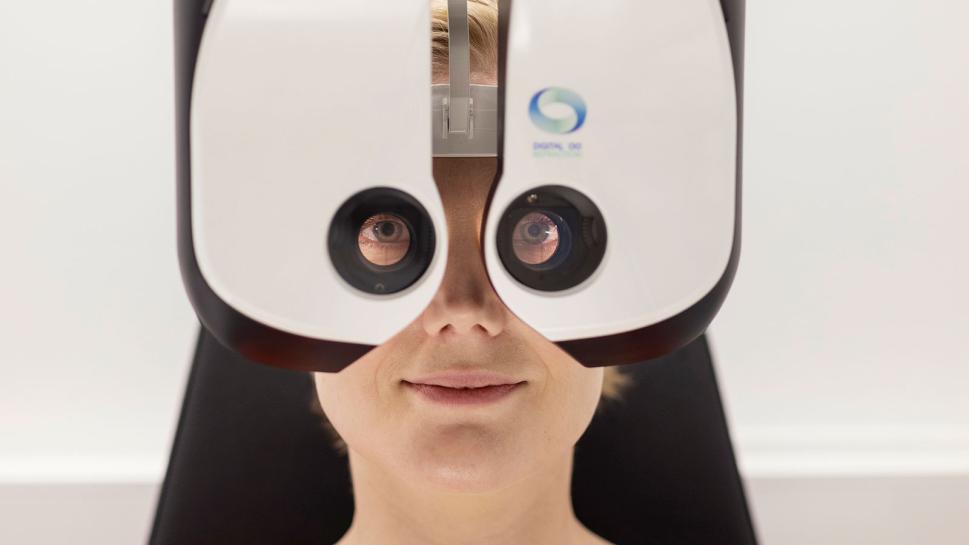
As our customer, you will have an access to various testing and experimentation environments in healthcare and social services: for example modern Simulation Hospital, Biomechanics and Movement Laboratory, Optometry Center, Oral Excellence and Onboard Metropolia Gerontechnology.
In addition to these facilities, you will have access to our expertise based on years of research, development, and innovation experience, tailored R&D services and the opportunity to utilize the multidisciplinary expertise of our students and Metropolia staff in developing innovations.
Welcome to learn more about our activities!
Contact us
We provide services in cooperation
Facilities for testing and research
Metropolia Proof Health consists of facilities operating on Metropolia's Myllypuro campus in Helsinki.
Metropolia Proof Health is a part of Health Proof Helsinki -ecosystem
Ethical principles
Metropolia is committed to the guidelines of the Finnish National Board on Research Integrity (TENK):
- Responsible conduct of research and procedures for handling allegations of misconduct in Finland. Guidelines of the Finnish Advisory Board on Research Integrity 2012
- The ethical principles of research with human participants and ethical review in the human sciences in Finland. Finnish National Board on Research Integrity TENK guidelines 2019
Metropolia is also committed to:
- Declaration for open science and research 2020-2025, Open Science Coordination in Finland, Federation of Finnish Learned Societies, 2020
- Ethical recommendations for thesis writing at universities of applied sciences, The Rectors’ Conference of Finnish Universities of Applied Sciences Arene, 2020
- Smart openness: 3UAS’s joint policy, promises and principles for an open operational culture, The strategic alliance of Haaga-Helia, Laurea and Metropolia, 2017
- Open Access statement by The Rectors’ Conference of Finnish Universities of Applied Sciences Arene, 2009
In international collaboration, Metropolia follows ALLEA’s (All European Academies) European Code of Conduct for Research Integrity (2017). (pdf)
In medical research involving human subjects, including research on identifiable human material and data, Metropolia follows the Declaration of Helsinki (World Medical Association, 1964, latest revision 2013.)
Research integrity, research ethics and the Health Testbed Excellence platform
In research or test activities carried out in the Metropolia Proof Health platform, the ethical questions focus on the interaction between the researcher and the research participant, as well as protecting the privacy and personal data versus openness of research (openness of process, research data and publications).
If there is any potential harm that may be caused to the people participating the research due to the research or its results, the project in question undergoes ethical review prior to gathering data. If the project is medical research as defined by law (Medical Research Act 488/1999), the ethical review is carried out by the ethics committee of medical research. In human sciences, an ethical review is carried out in The Human Sciences Ethics Committee of the Helsinki Region Universities of Applied Sciences. Th
Research participants are informed if the research data will be opened (what data, if it is anonymous or pseudonymous, and where it will be archived etc.) Openness of research data is considered at the planning stage of each project or test activity. The data management plan, containing a description of the processing of personal data throughout the lifespan of the project, is made with DMPTuulionline tool. There are guidelines, DMPworkshops, data support, and a model of data management available at Metropolia. When collecting, preserving and opening research data, only reliable services and software used.
The protection of the research participants privacy and people who have provided information for the research, must also be considered in research publications. Decisions are made on a case-by-case basis.
Ensuring the safety and performance of medical devices is highly regulated. The Medical Device Directives set out procedures to be followed by national authorities.
Finnish Medicines Agency Fimea
European Commission Public Health Directives
Openness of research
Metropolia is committed to the Open Access statement by The Rectors’ Conference of Finnish Universities of Applied Sciences Arene, which means that all scientific articles written by personnel of Metropolia should simultaneously published in the Open Repository, Theseus.